Watch how easy is it to use Apollo!
General Apollo FAQ
Please refer to the Apollo user manual here: https://inbio.com/Apollo-User-Manual
You can purchase new filters from our store here: https://store.inbio.com/apollo-air-sampler/Apollo-Air-Sampler-Replacement-Filters-10x
Should particulates build up on the device, we recommend the use of compressed air to remove the dust and loose residue from the device. Detergent based wipes and cotton swabs can be used to clean the front and in-between the grills should residue remain. After cleaning, the device should be switched on and run for 10 minutes with the filter holder removed, to allow any loose particulates to be removed. The filter holder can be cleaned with standard detergent, wiped and should be allowed to fully dry before reinserting into the device. Do not immerse the Apollo in water or other liquids.
For microbial applications, please see relevant FAQ below.
Rental options are available for our Apollo devices, depending on the application. Please get in contact with Ross Yarham (Ryarham@inbio.com) and provide your intended use and planned duration of rental.
Industrial and personal air sampling devices are well known for being noisy and cumbersome. Apollo has been specially developed to remove this inconvenience from air sampling and whilst running, it is no louder than ~40 decibels or the equivalent to a quiet library!
The Apollo device, using our specialised allergen capture filter, has an average air flow rate of 540L/min.
Apollo for Allergens FAQ
Traditional methods for aerosolised allergen sampling include Institute of Occupational Medicine (IOM) samplers and Electrostatic Dust Collectors (EDC). IOMs are designed for personal sampling using a specialized pump, which require calibration, and are loud/cumbersome to use. EDCs only allow for passive collection of settled dust.
When designing Apollo, InBio aimed to enhance the user experience while improving the sampling methods available to the IAQ industry. The result is a quiet, high throughput air sampler which allows for efficient, easy, and reproducible allergen sampling. Compared to traditional air sampling methods, Apollo demonstrates improved sensitivity and collects up to 10x more allergen across a range of indoor and food allergens – and is as simple as plugging in!
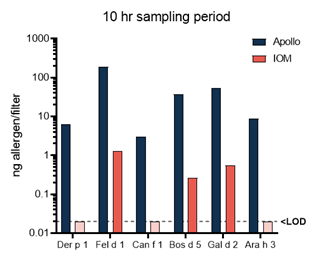
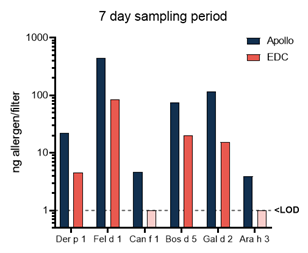
Figure 1 – Representative data from 1 household. IOM and Apollo devices were run simultaneously for 10 hours and EDC and Apollo for 7 days. Results are provided in ng allergen per filter on a log scale.
There are some slight differences in terms of reporting limits between the Apollo Ambient Air sampler and IOM devices. An example scenario has been laid out below to demonstrate.
If you were measuring levels of mouse (Mus m 1) by our InBio’s MARIA assay, the limit of detection (LOD) of that assay is currently 0.01ng/ml.
- For IOMs, the sample extraction would be done in 1ml of assay buffer, therefore the LOD for that result would be 0.01ng/ml = 0.01ng/filter.
- For the Apollo, we would do the extraction in 10mls, therefore the LOD for that would be 0.01ng/ml = 0.1ng/filter. This however does not take into account sampling time.
The preferred units for exposure assessment results is in ng/m3. This is where the Apollo is more sensitive in terms of LOD based on sampling time, and hence in terms of ng/m3.
If we take an example where an air sample was taken for 1 hour:
- With the IOM pump set to 2L/minute, that would lead to 120L or 0.12m3 of air being sampled. With the LOD of the assay at 0.01ng/filter, the LOD would then be 0.083ng/m3.
- With the Apollo pulling ~540L/minute, that would lead to 32,400L or 32.4m3 of air being sampled. With the LOD of the assay at 0.1ng/filter, the LOD would then be 0.003ng/m3 (~27x lower).
The Apollo can be extremely beneficial to occupational exposure assessments.
For personal monitoring, traditional IH practices, such as IOM samplers still provide a robust indicator of what an individual is exposed to per specific task. However, collecting air samples from stationary areas or larger rooms using these techniques is not suitable and can often report false negatives, whereas the Apollo has been designed specifically to sample large volumes of air from whole rooms. A particular example would be rooms where there is supposed to be low animal handling activity and consequently low allergen levels. This would be an ideal place for Apollo sampling to take place.
We have conducted trials using the Apollo in experimental animal facilities in the UK. The Apollo was placed in ‘clean’ rooms where no allergen should have been present, however it was suspected that H&S protocols were not being strictly adhered to and consequently it was supposed that allergens were being transferred from ‘dirty’ rooms to ‘clean’ rooms. Identification of lab animal allergens in these rooms by the Apollo confirmed that allergen was present in rooms it should not have been.
In laboratory animal facilities, the Apollo can be employed for very short durations in rooms where allergens are expected to be high, such as rooms housing animals in conventional or filter top cages or where tasks such as cage cleaning or washing occur. Allergen will be picked up with short duration sampling time (less than 1 hour) in these situations. In rooms where lower allergen is thought to be present, longer duration sampling can be employed. Allergens have been picked up in ‘clean’ rooms with as short a sampling time as 2.5 hours. However, the Apollo is also suitable for much longer duration of anywhere between 24hrs to 7 days.
See Submitting Samples.
Please complete and provide InBio’s analysis request form, which is to be included with the sample shipment.
Pleas include the sampling duration under the special instruction column.
Standard turnaround time from sample receipt is five business days for the US lab, and ten business days for the UK lab.
Please contact us for detailed instructions.
Briefly;
1) Place the filter back into a ziplock bag or a suitable container.
2) Add 10mls extraction buffer (e.g. PBS-Tween 0.05%)* to the bag/container and seal.
3) Extract for 2 hours at room temperature with gentle agitation (laboratory rocker or shaking incubator).
4) Remove supernatant with a pipette and store extract until allergen measurement can be performed.
5) Use InBio’s immunoassays for accurate measurement of allergens.
Please also see our other FAQs for common questions on our ELISA 2.0 and MARIA assays.
*alternative extraction buffers can be used, contact InBio for any queries.
Our Apollo, using our specialised filter, can capture a vast range of airborne food and environmental allergens, including but not limited to peanut, milk, egg, tree nuts, cat, dog, rat, mouse, dust mite and pollen.
Learn more about the Apollo here: https://inbio.com/wp-content/uploads/2022/10/Development-of-the-Apollo-a-novel-static-air-sampler-for-the-measurement-of-airborne-allergen-exposure_.pdf
Sufficient sampling time will vary dependant on the application and setting. In most settings, for initial screening for airborne contaminants we would recommend a sampling time of around 12-24 hours to allow for a high volumes of air to be sampled. Please note the duration of sampling on the sample submission forms.
Sampling duration examples*:
- Home & domestics environments – 12-24 hours
- Office & Commercial environments – 1-5 days
- Animal research facilities/houses – 1-8 hours (task dependant)
*Situations may vary, please contact us to discuss your specific scenario & environment.
After sampling for allergens, the filter should be stored sealed in a clean ziplocked bag or other suitable container and refrigerated (2-8°C) before being sent to InBio for analysis or analysed in your own laboratory using InBio’s immunoassays.
If it is not possible to frigerate the sample, then it should be sent to InBio for analysis or analysed in your own lab within one week of collection.
Our Apollo devices are suitable for the detection of endotoxin in indoor environments.
Our data demonstrates detectable levels of endotoxin from as little as 1 day, and that increased sampling time correlates linearly with endotoxin concentration up to at least 7 days.
InBio are currently undertaking research to establish & optimise Apollo’s uses to capture a range of microorganisms, including fungi/molds, bacteria and bacteriophages. Early proof of principle experiments conducted have suggested good efficacy.
If you’d like to be kept up to date with our developments just get in touch to learn more and discuss your potential applications.
Apollo for Microbes FAQ
Apollo is capable of capturing airborne bacteria and viruses in indoor environments.

Our data demonstrates that Apollo can collect high numbers of viable bacteria and bacteriophage with short duration sampling. In addition, when sampling is coupled with qPCR detection, sampling time can be increased and higher numbers of bacteria can be collected.

Figure 1. Overview of microbes which Apollo has been shown to capture. Representative data for Apollo measurement of S. aureus, K. pneumoniae and E. coli within a test chamber (Viable N=2-4, qPCR N=1). Results provided as bacteria/m3 on a log scale.
The sampling times and filter extraction methods may be different for measuring airborne microbes, compared to measuring airborne allergens or endotoxin, depending on the application.
Sampling time will vary depending on the desired application and whether measuring the number of viable or total microorganisms present.
Similarly, the filter extraction process will be application dependent.
Currently we do not offer an option to return microbial samples for routine analysis at InBio, but we are exploring this capability – watch this space!
However, we are happy to provide recommendations to support you with your microbial sampling and analysis and to explore custom analysis options at our labs in the UK.
InBio’s data demonstrates that Apollo is suitable for detection of airborne β-glucans, which are often used as surrogates for Mold exposure. These molecules are known to be immunomodulators, and airborne exposure is linked with respiratory symptoms.
β-glucans are derived from the cell walls of most fungi and some bacteria and plants. They are frequently found in indoor environments and can become airborne.

Figure 1. Measurement of airborne Beta glucans and Endotoxin. Data from sampling in homes with Apollo for 7-days. Beta glucans quantified by Glucatell assay and Endotoxins quantified by Endozyme ® II rFC assay.
InBio’s Apollo data shows no correlation between β-glucan levels and Endotoxin (derived from Gram-negative bacteria) within the homes sampled. This result is similar to previous observations by Milton et al. 2001 and is likely reflective of the different microbial sources that endotoxin and β-glucan are derived from.
We recommend cleaning the Apollo and filter holder with a laboratory grade surface disinfectant* that has proven efficacy against a broad spectrum of microbes, e.g., 1% Chemgene™.
The filter holder can be removed from the device and sprayed with the chosen disinfectant and left for the required contact time, before drying with blue roll or similar. The Apollo device should be wiped with the disinfectant, including the grills. Following wiping with the disinfectant, we recommend wiping the device with the 70% Isopropanol.
If there is a concern with DNA carry over between sampling in highly sensitive molecular applications, an additional step could be included to wipe Apollo with DNA AWAY™ or a similar product.
Do not spray disinfectants directly on to the grills/fan. Do not immerse the Apollo in water or other liquids.
*Check material compatibility of disinfectants before using to clean Apollo.

Have any other questions?