Please contact us.
- EPC-DP1: Uses capture mAb 5H8 and biotinylated mAb 4C1 for detection
- EPC-DP1A: Uses capture mAb 10B9 and biotinylated mAb 5H8 for detection
EPC-DP1 (5H8/4C1) is the most widely used ELISA kit. The biotin 4C1 mAb is also used in the Der f 1 ELISA (EPC-DF1). EPC-DP1 is recommended for most routine analyses of dust and air samples for Der p 1. However, there is a degree of cross-reactivity for Der f 1 in the EPC-DP1 assay, of about 2% (see Luczynska et al, J Immunol Meth, 1989). For routine analyses, this level of cross-reactivity is not significant.
The EPC-DP1A kit shows <0.1% cross-reactivity with Der f 1 and is designed for applications that need absolute distinction between Der p 1 and Der f 1. For example, an allergen manufacturer who wants to be sure that they have no cross contamination between species in their dust mite cultures. The EPC-DP1A assay has the same level of sensitivity as the EPC-DP1 kit (1-2ng/ml).
InBio markets two ELISA kits to measure mite group 2, which use the same combination of monoclonal antibodies in both assays, but different standards:
- EPC-DP2: Uses capture mAb 1D8 and biotinylated mAb 7A1 for detection with a Der p 2 standard
- EPC-DF2: Uses capture mAb 1D8 and biotinylated mAb 7A1 for detection with a Der f 2 standard
When testing a mixed mite culture, the concentration measured by each assay is not directly proportional to the amount of dust mite culture (Der p v Der f).
Data previously measured by the EL-MG2/EPC-D2 assay used the Der p 2 standard, which is the same standard included in the EPC-DP2 kits.
InBio ELISA kits contain sufficient reagents (capture mAb, biotinylated detector mAb and allergen standard) for the number of plates included in the kit. The number of samples that can be analyzed per plate depends on the number of dilutions used for each sample.
Standard curve in duplicate, with 4 blank (PBS-T) wells, uses 24 microtiter wells, leaving 72 wells for samples on each plate. Typically, we use 4 doubling dilutions for each dust sample (1/10, 1/20, 1/40, 1/80 for mite allergens), which means that 18 samples can be tested on a plate.
Air samples usually contain low levels of allergen and are assayed at 1/2, 1/4, 1/8, and 1/16 dilution, which means that 18 samples can be tested on a plate.
Typically, we use a minimum of 6 doubling dilutions for each food sample (1/1, 1/10, 1/100, 1/1000, 1/10,000, 1/100,000), which means that 12 samples can be tested on a plate.
We use a minimum of four dilutions per sample so that we can in most cases obtain results on >90% of the samples in a single assay. More dilutions increase the probability of having at least two points on the linear part of the control curve for each sample. It may be possible to use fewer dilutions, if the samples are known to have allergen levels in a particular range. Other samples, e.g. allergenic extracts or food source materials, may have very high allergen levels and have to be assayed at doubling dilutions starting at 1/100 or 1/1000, and may need to be serially diluted across the ELISA plate.
We routinely keep house dust samples at 4°C prior to assay, but once extracted they should be stored at -20°C.
We routinely keep dry food samples at 4°C prior to assay, but once extracted they should be stored at -20°C.
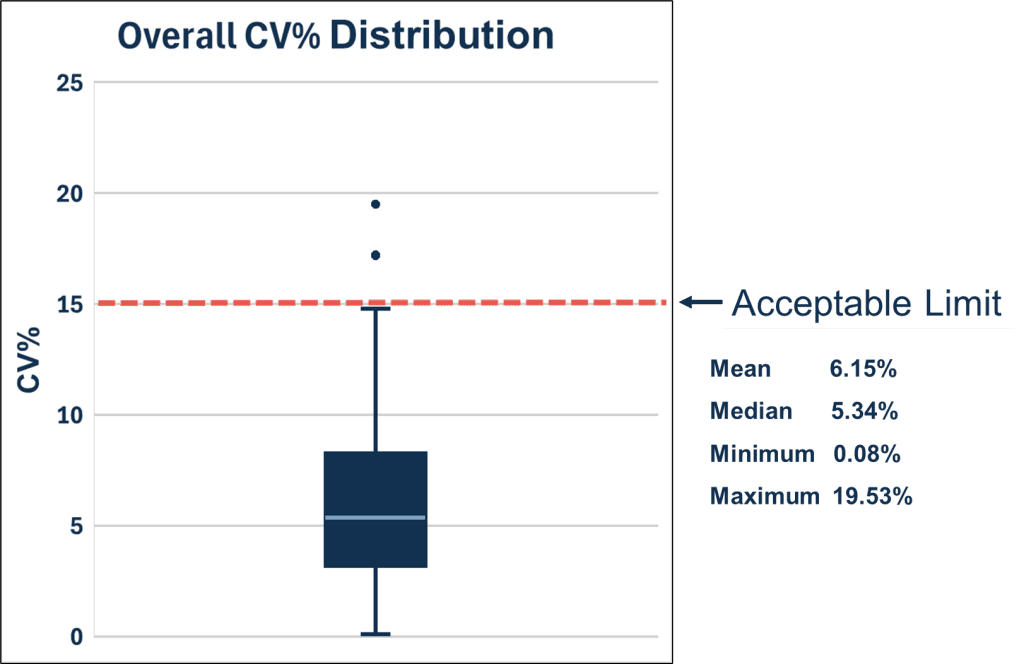
InBio has performed internal stress tests on ELISA 2.0 kits (n=11 allergens) to simulate the conditions of a delayed shipment. All ELISA 2.0 components were left at room temperature protected from light for 7 days (average temperature was 78°F/25.5°C). The ELISA 2.0 kits were run in parallel to kits that remained at the recommended storage temperature (40°F/4°C). Sample (n=54) results between plate conditions were compared and no negative effects were observed. The average coefficient of variation (CV) of all samples for 11 allergens is depicted in the box plot below. The majority of data falls under an acceptable limit of 15%, which is below the possible assay variability of 20%.
No, it is important to store the pre-coated plates at 4°C until use and protected from exposure to humidity in the air. We do not recommend the partial use of a pre-coated plate. In addition, the supplied reagent volumes are optimized for the number of plates included in the kit.
All steps are carried out at room temperature, usually 20-25°C. Plates and reagents should be brought up to room temperature prior to using.
We noticed that the assay was very slow in developing (and never reached OD 2.0). Is this a problem?
Here are a few things you can check:
- Have the biotinylated reagents been frozen at any time during storage? This may reduce the sensitivity of those mAbs.
- Are you making up the reagents or using buffer tablets? We’ve found that some buffer tablets do not work as well as fresh-made solutions. We usually keep all solutions for less than a month at 4°C without Thimerosal.
- Has sodium azide been used in any of the reagents as preservatives? This would stop the color development since it is an inhibitor of horseradish peroxidase.
- Has your TMB been exposed to light for a significant period of time? The color will turn to blue the longer it is at room temperature and exposed to light.
- Did you add the stop solution after the OD reached 0.8-0.9?
- Plate washer contamination can sometimes hinder color development causing the plate to develop very slowly or never reaching an O.D. of 2.0. Plate washers should be cleaned on a regular basis.
- Direct sunlight exposure via windows will cause both of the above issues. Place ELISA plates away from windows during incubation.
Reduce the amount of streptavidin-peroxidase or goat anti-rabbit added during the detection antibody step. Start by reducing the amount to 2/3 the volume listed in the CoA without changing anything else. If the plate develops too quickly after this adjustment, reduce the volume to half the volume listed in the CoA.
This is an indication that the plate developed for too long prior to stopping the reaction with the provided stop solution. An S1 450nm OD of 3.5 or greater after TMB addition is at risk of developing black precipitate in the wells over time. To avoid this issue, either stop the plate when the OD for S1 is around 0.08 to 0.09 (prior to adding stop solution) and/or read the plate immediately after stopping development. All plates should be read within 10 minutes of adding the stop solution.
After adding the stop solution, you should read the plates within 30 minutes.
The background on the assay is usually higher in assays where the secondary antibody is a rabbit polyclonal. The background is usually around 0.15 OD. Other causes of the high background that you can check:
If washing plates by hand, an increase in washes throughout the assay can help lower the background. Fill the wells completely without overflow and then dump the wells carefully not to contaminate. Then gently tap the excess buffer from the plate on a paper towel.
After adding the TMB, shake gently by tapping the plate on the side occasionally and right before reading to ensure the color development is homogeneous throughout. Repeat the same process after adding the stop solution.
Pipetting is very important when running the ELISA assays. Be sure that the pipettes being used, especially multichannels, are picking up and dispensing evenly during your doubling dilutions across the plate. Also be sure that there are no air bubbles during your transfers.
Ensure starting concentration is correct; i.e. for ELISA 2.0, using 20uL standard in 180uL assay buffer as standard 1 dilution and doubling dilutions for a 10-point standard curve. Standard curves should be run in duplicate. Also, assay development time and maximum OD can change the shape of the standard curve.
Ensure all reagents and buffers are at room temperature. Use a plate cover or sealer, plate rocker (gentle agitation), and/or isolate the plate(s) from environmental temperature inconsistencies by using an incubator set at 20°-25° Celsius. If running multiple plates at once, avoid stacking to ensure even temperature across each plate.
If you don’t find your answer in the questions listed above, please submit a troubleshooting form to support@inbio.com that includes specifics about your issue, kit details, plate data and/or your data file.